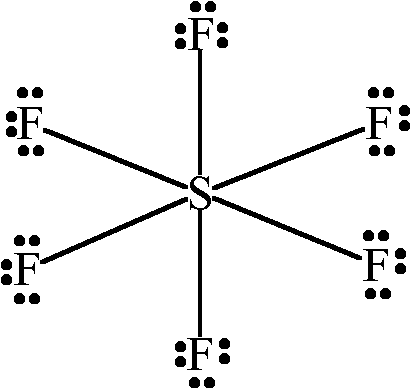
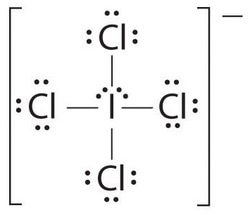
Octahedral
The Octahedral shape is a type of shape which a molecule takes form of when there are 6 bonds attached to a central atom with 4 on the same plane. There are no lone pairs attached to it. The bond angle between the bonds is exactly 90 degrees. The molecule is non-polar since it is a symmetrical shape. Here are some examples of Octahedral-shaped molecules:
Sulfur Hexafluoride
What contributes to this shape?
The only thing that contributes to this shape are the six bonds which the central atom is attached to. There are no lone pairs of electrons which would repel with the surrounding atoms and provide a different bond angle and shape. Since there are only 6 bonds attached to it and no lone pairs, the bond angles tend to be 90 degrees, meaning the bonds won't bend.